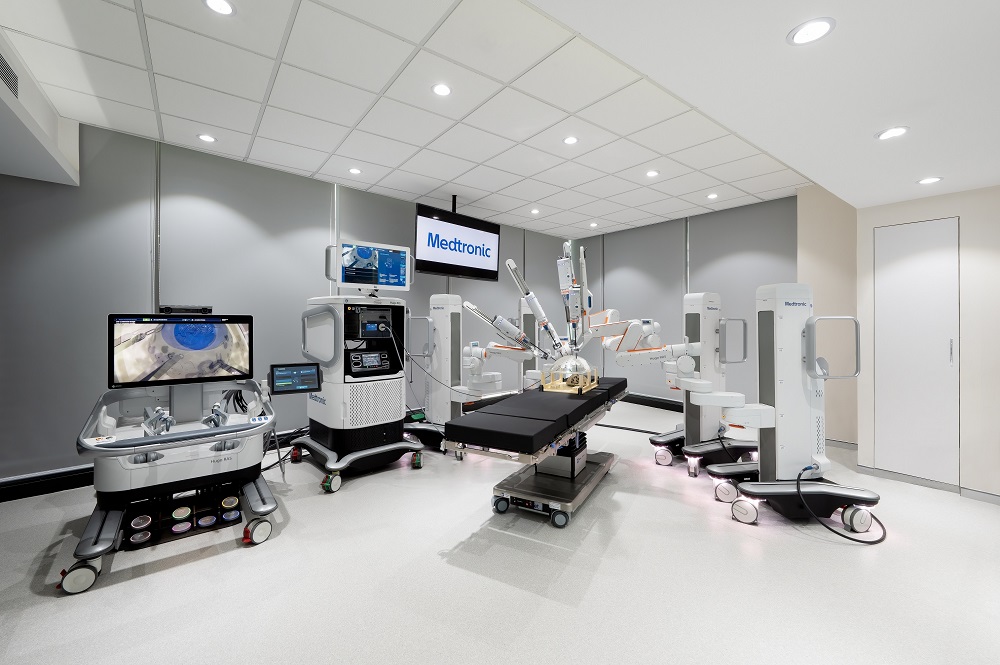
Medtronic Showcases Hugo™ Robotic-Assisted Surgery System At The Inauguration Of Its First Surgical Robotics Experience Center In India
- Hugo™ RAS System to Help Advance Capabilities in Minimally Invasive Surgery and Expand Access to Care
- The Surgical Robotics Experience Center, First in APAC, Will Enable Clinicians in India to Experience and Train on Robotic-Assisted Surgery Technology from Medtronic
Bengaluru: India Medtronic Private Limited, a wholly-owned subsidiary of Medtronic plc (NYSE: MDT) – the world’s largest medical technology, services, and solutions company – today showcased its new robotic-assisted surgery platform, Hugo™ RAS system, at its state-of-the-art Surgical Robotics Experience Center (SREC), which was inaugurated in Gurugram. The center was inaugurated by Padma Bhushan Dr. BK Rao, Chairman NABH, Chairman ASSOCHAM National Health Committee and Former Chairman- Board of Management at Sir Ganga Ram Hospital, Delhi in the presence of Chief Guest Shri Amitabh Kant, CEO, NITI Aayog. Committed to delivering solutions that help surgeons and operating room staff achieve procedural and clinical proficiency, the SREC will focus on standardized training for surgeries with the Hugo RAS system. The SREC is the company’s first in Asia Pacific and one of ten world-class facilities around the globe today where clinicians can experience and train on the Hugo RAS system.
The Hugo RAS system — Medtronic’s solution to historic cost and utilization barriers that have kept surgical robotics out of reach for many hospitals — is a modular, multi-quadrant platform designed for a broad range of soft-tissue procedures. With the Hugo RAS system, Medtronic is building on its decades-long commitment to bring the benefits of minimally invasive surgery (MIS) to more patients around the world. With MIS, patients experience fewer complications, shorter hospital stays, and faster return to normal activities.1-3,†
Reiterating the need for pathbreaking medical technology for India, Shri Amitabh Kant, CEO, NITI Aayog stated, “While we believe promoting innovation in healthcare is crucial to building a healthy nation, it is also critical to invest in continuous training and education of the healthcare personnel. The opening of Medtronic’s Surgical Robotics Experience Center is a befitting example of achieving holistic healthcare goals in terms of accessibility and training so that more patients can benefit from latest advancements in med-tech.”
During the inauguration, Padma Bhushan Dr. BK Rao, Chairman NABH, Chairman ASSOCHAM National Health Committee and Former Chairman- Board of Management at Sir Ganga Ram Hospital, Delhi echoed the contribution of Medtronic in transforming healthcare in India and said, “Innovation is the cornerstone of medical technology advancement and towards saving lives. The advent of robotic surgeries in India is a hallmark of the progress we’ve made in improving access to quality healthcare for patients. I am confident that Medtronic’s contribution through Surgical Robotics Experience Center will help in training surgeons and in improving adoption of minimally invasive way of surgery.”
Through SREC, the aim is to provide a multi-layered curriculum for training surgeons, assistants, and paramedical staff on the Hugo RAS system in India. The curriculum includes training such as dry lab surgical models, simulation exercises, and robotic instruments and accessories to provide a focused and collaborative experience.
“Training and education of surgeons is an important driver in accelerating medical technology adoption and improving access to healthcare. I believe in a way it is aligned with Government of India’s focus on Skill India. Through the Surgical Robotics Experience Center, we are dedicated to helping surgeons focus on deepening their expertise in robotic-assisted surgery,” said Madan Krishnan, vice president and managing director, Medtronic India. “The Hugo RAS system demonstrates our commitment to the rapidly changing surgical environment by improving experience for our surgeons while delivering quality care to patients.”
“We designed the Hugo RAS system to bring the benefits of minimally invasive surgery to more patients in more places around the world, and it’s incredibly exciting that India is leading the way forward for this new era in robotic surgery,” said Megan Rosengarten, president of the Surgical Robotics business, which is part of the Medical Surgical Portfolio at Medtronic.
“India has a vast number of extremely qualified surgeons and hospitals that nurture healthcare solutions for the patients who need them. SREC has been set up with a vision to provide healthcare professionals a complete hands-on experience of our new surgical robotics system,” said Kevin Falzon, senior business director of Surgical Robotics Asia Pacific, who attended the inauguration virtually. “We’ll be training surgeons on the use of the Hugo™ RAS system, utilizing procedural and evidence-based medical education activities. The center is also equipped with mixed reality technology to provide remote virtual tours to clinicians, administrators, and OR staff.”
The Hugo RAS system combines the precision of wristed instruments with the flexibility of independent robotic arm carts and best-in-class 3D visualization technology on an open console. Touch Surgery™ Enterprise, a cloud-based surgical video capture option powered by artificial intelligence, allows surgeons to seamlessly record, analyze, and share Hugo system case footage.
The Medtronic Hugo RAS System is commercially available in certain geographies. Regulatory requirements of individual countries and regions will determine approval, clearance, or market availability. Hugo RAS is pending CE mark for the EU. In the U.S., the Hugo system is an investigational device and is not for sale. Touch Surgery Enterprise is not intended to direct surgery, or aid in diagnosis or treatment of a disease or condition.